
What is a Calibration Certificate or Calibration Record as per ISO17025?

|
In this article, you will learn what is a calibration certificate according to the FDA requirements. Calibration records are commonly known as calibration certificates contain the equipment identification, calibration dates, the individual performing each calibration, and the next calibration date. The following picture shows an example of a calibration certificate for a temperature and relative humidity data logger sensor. It contains the most typical information as certificate num, instrument name, equipment model, serial number, manufacturer, calibration date, calibration expiration date, instrument condition, calibration standard reference, etc. 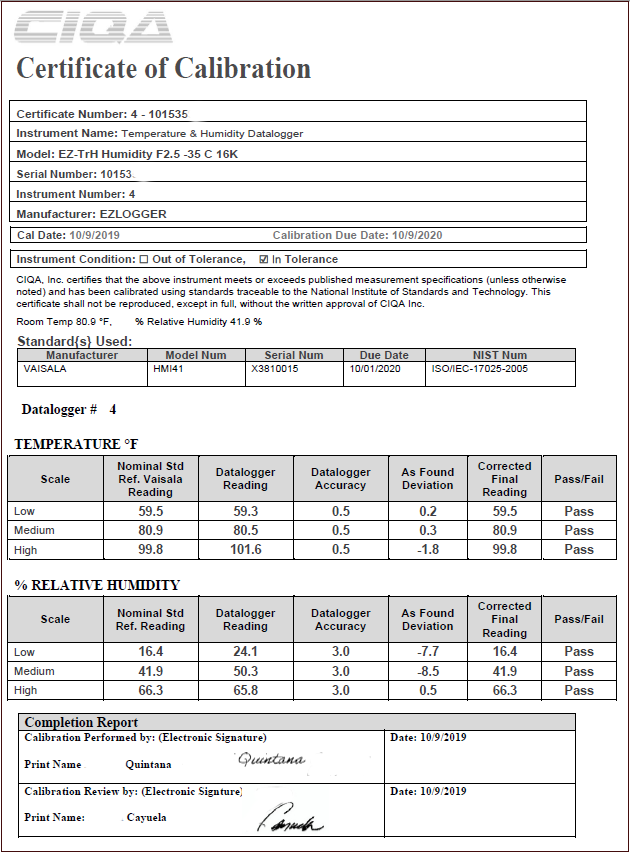
In addition to the calibration certificate, a calibration tag or label is placed or displayed on or near each piece of equipment or shall be readily available to the personnel using such equipment and to the individuals responsible for calibrating the equipment. 
In most cases the calibration tags are placed in front of the equipment or gauge and contain the following information:

CIQA is a quality and regulatory consultant with 25 years of experience developing products and managing projects in the medical device supply chain, and pharmaceutical industries. His experience includes research, product development, operations management, manufacturing engineering, equipment design, regulatory affairs, and quality assurance. Specific questions about Document Control Management DCM or quality system training can be directed to CIQA at ramon.cayuela@ciqa.net. SUBSCRIBE AND FOLLOW US TO LEARN MORE.For more details about what is a calibration certificate, follow us.Three (3) Options to Create Document Control Management DCM Procedures:Bronze Option. You Can Create Your Own Quality Procedures, using a Template.You can download samples of the Document Control Management DCM procedure templates in .pdf format. To see the complete list of the most popular quality procedures templates, click here. In addition, you can request a quotation to buy online a full SOP template document in MS Word format that is completely editable, ready to fill, and adapt to your specific needs. Silver Option. We Can Bring You a Formal Training about what is a calibration certificate.This option is recommended if you want to learn more about how to build robust quality system procedures. One of our expert(s) can provide online step-by-step training to your team (unlimited assistance) on how to build reliable SOPs using our template(s). Also, you can improve your corporate quality procedures and policies by incorporating our template(s) and tools. It includes a fully editable template from the Bronze option, plus training, exams, and a training certificate for each assistant. Request a quote now. Gold Option. We Can Create Customized Quality Procedures.One of our expert(s) will create and prepare your customized SOPs with the inputs and specific information of your company. It includes a fully editable template from the Bronze option, plus online support in document creation, implementation, and training. Request a quote online. Get in compliance with what is a calibration certificate, CONTACT US (Hablamos Español)REFERENCES:For more information about what is a calibration certificate, refer to:STATUTORY AND REGULATORY REQUIREMENTS TITLE 21–FOOD AND DRUGS PART 820 — QUALITY SYSTEM REGULATION Subpart G–Production and Process Controls Sec. 820.72 Inspection, measuring, and test equipment. httpss://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=820.72 ISO/IEC 17025 TESTING AND CALIBRATION LABORATORIES ISO/IEC 17025:2017 specifies the general requirements for the competence, impartiality and consistent operation of laboratories. ISO/IEC 17025:2017 is applicable to all organizations performing laboratory activities, regardless of the number of personnel. httpss://www.iso.org/ISO-IEC-17025-testing-and-calibration-laboratories.html TITLE 21–FOOD AND DRUGS PART 111 — CURRENT GOOD MANUFACTURING PRACTICE IN MANUFACTURING, PACKAGING, LABELING, OR HOLDING OPERATIONS FOR DIETARY SUPPLEMENTS – What is a Calibration CertificateSubpart D–Equipment and Utensils httpss://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=111.35 21 CFR § 58.63 – Maintenance and calibration of equipment.
httpss://www.law.cornell.edu/cfr/text/21/58.63 40 CFR § 160.63 – Maintenance and calibration of equipment.
httpss://www.law.cornell.edu/cfr/text/40/160.63 For more information about what is a calibration certificate, refer to:httpss://whatis.techtarget.com/definition/calibration-management-software |

Ramon Cayuela, MS, BS, Chemical Engineering
CIQA President and CEO.
I've been working in validation engineering since 1992 with many multinational pharmaceutical companies. I love sharing my passion and knowledge with others. If you have any questions about anything (or just have general questions). I will be more than happy to assist you. You can count on the BEST customer service on CIQA. I go to great lengths to make sure my clients are 100% satisfied with their purchases and check emails/messages consistently throughout the day. You can rest assured that everything being sold here is as-described or your money back. I look forward to working with you!
Related Articles
Subscribe to get validation
news and free tips by email.
Need Additional Help?















